 
Water or oil will tend to be quite happy away from the interface, reducing its The reason why 90° is optimal is that particles that are too easily wetted by Other half that is hydrophobic, though these are not so easy to produce. The ideal is a Janus particle with one half that is hydrophilic and the In general particles with a 90° water contact angle seem to be the best all-roundĮmulsifiers. 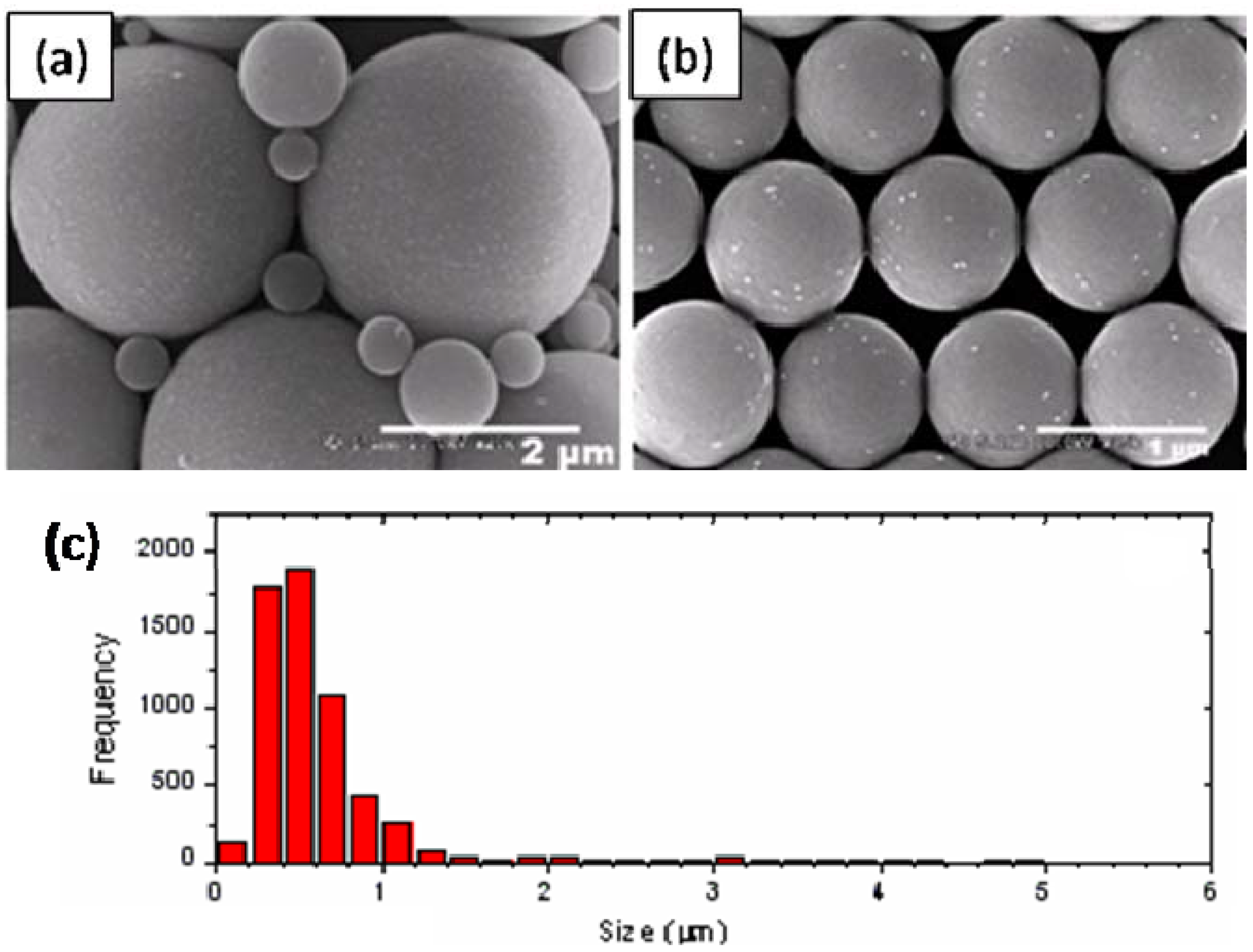
O/W emulsion and intermediate oleophilicity tends to give W/O emulsions. Intermediate hydrophilicity tends to give an If the particles’ surface isĮasily wetted by oil or water they are of no use as they will spend most of their Their design rules are straightforward in principle. These particles probably partition too much into their preferred phase so would be inefficient emulsifiers. (W Ramsden found them four years before S Pickering), but they are commonlyįigure 4‑12 If the particle favours water then the emulsion is O/W, if the particle favours oil then the emulsion is W/O. To create a rather stable shell around the internal drop. The logical continuation of this trend is to have solid particles that can assemble Too insoluble in the water phase to allow any Ostwald ripening. Is a chance to coalesce – and they provide big steric barriers, and/or they are Large proteins or phospholipids are too slow to move out of the way when there 
On the other hand, once they’veįormed an emulsion they tend to be stable over longer timescales because the Good at producing low interfacial energies. This is bad when you want the emulsionĪ lot of food science emulsions are made with relatively “bad” surfactants thatĪre slow to reach any interface and often pack so poorly that they aren’t too Interface in order to create an emulsion. This is good when you want them to come quickly to the Thermally responsive emulsions were created with the SiO 2−PDMAEMA particles such that stable emulsions prepared at low temperature were rapidly broken by increasing the temperature above the CFT.Classical emulsions are made using small-molecule surfactants that are Emulsions have been stable for over 13 months, and some have dispersed as much as 83 vol % oil in the emulsion phase. Both good (xylene) and poor (cyclohexane) solvents could be emulsified, but the poor solvent could be emulsified over a broader range of conditions than the good solvent. The lowest grafting density particles (0.077 chains/nm 2) proved to be the most efficient and robust emulsifiers, producing stable emulsions using as little as 0.05 wt % particles in the aqueous phase and successfully emulsifying over a broader range of solution conditions than for the higher grafting density particles (0.36 and 1.27 chain/nm 2). The preferred emulsion type was oil in water in all cases. The effects of the solvent quality of the “oil” for the PDMAEMA brush were studied in addition to the effects of aqueous pH, ionic strength, and temperature relative to the CFT. ATRP provides a high degree of control over the brush grafting density and degree of polymerization, two of the principal variables examined in this study. Accordingly, SiO 2−PDMAEMA nanoparticles were thermally responsive, as shown by the fact that they displayed a critical flocculation temperature (CFT) when heated. PDMAEMA is a water-soluble weak polyelectrolyte with a pH-dependent lower critical solution temperature (LCST). The grafted nanoparticles were used to stabilize xylene-in-water and cyclohexane-in-water Pickering emulsions. A study is presented of emulsification by silica nanoparticles with poly(2-(dimethylamino)ethyl methacrylate) brushes grafted from their surfaces (SiO 2−PDMAEMA) by atom-transfer radical polymerization (ATRP). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
